 
The distinction between metals and nonmetals is one of the most fundamental we can make in categorizing the elements and predicting their chemical behavior. 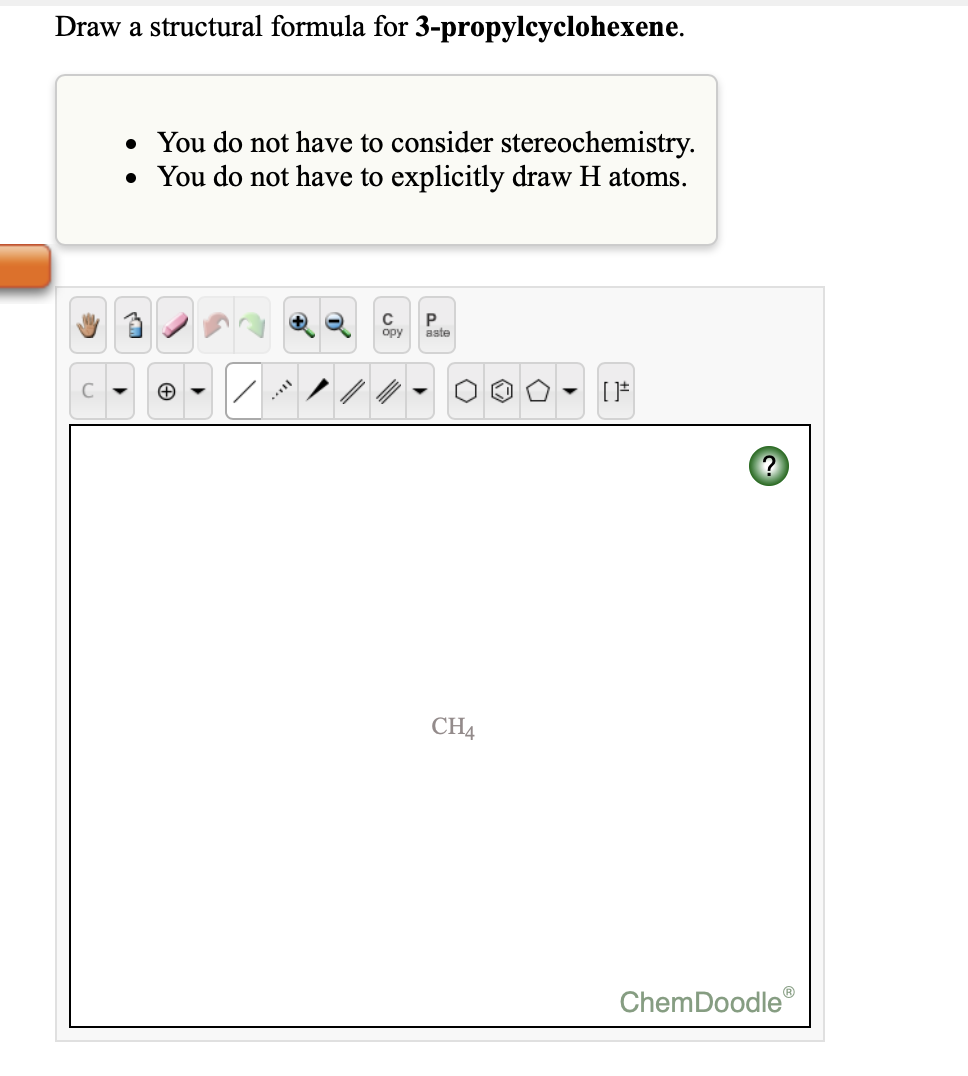
These are the semimetals (or metalloids), elements that have some of the chemical properties of both nonmetals and metals. In between the metals and nonmetals, along the heavy diagonal line running from B to At is a group of elements with intermediate electronegativities (χ ~ 2.0). In contrast, elements with a low electronegativity (\(\chi \le 1.8\)) have electron affinities that have either positive or small negative values and small ionization potentials, so they are generally metals and good electrical conductors that tend to lose their valence electrons in chemical reactions (i.e., they are reductants). Elements with a high electronegativity (χ ≥ 2.2 in Figure 2.12.2) have very negative affinities and large ionization potentials, so they are generally nonmetals and electrical insulators that tend to gain electrons in chemical reactions (i.e., they are oxidants). Because electronegativities generally increase diagonally from the lower left to the upper right of the periodic table, elements lying on diagonal lines running from upper left to lower right tend to have comparable values (e.g., O and Cl and N, S, and Br).Įlectronegativity Differences between Metals and NonmetalsĪn element’s electronegativity provides us with a single value that we can use to characterize the chemistry of an element. If we ignore the inert gases and elements for which no stable isotopes are known, we see that fluorine (\(\chi = 3.98\)) is the most electronegative element and cesium is the least electronegative nonradioactive element (\(\chi = 0.79\)). Periodic variations in Pauling’s electronegativity values are illustrated in Figure 2.12.1 and Figure 2.12.2. Figure 2.12.1 A Plot of Periodic Variation of Electronegativity with Atomic Number for the First Six Rows of the Periodic Table Pauling arbitrarily set the electronegativity of fluorine at 4.0 (although today it has been refined to 3.98), thereby creating a scale in which all elements have values between 0 and 4.0. The original electronegativity scale, developed in the 1930s by Linus Pauling (1901– 1994) was based on measurements of the strengths of covalent bonds between different elements. 
For example, all scales predict that fluorine has the highest electronegativity and cesium the lowest of the stable elements, which suggests that all the methods are measuring the same fundamental property. Nevertheless, when different methods for measuring the electronegativity of an atom are compared, they all tend to assign similar relative values to a given element. In fact, an atom’s electronegativity should depend to some extent on its chemical environment because the properties of an atom are influenced by its neighbors in a chemical compound. Unlike ionization energy or electron affinity, the electronegativity of an atom is not a simple, fixed property that can be directly measured in a single experiment. Elements with low electronegativities tend to lose electrons in chemical reactions and are found in the lower left corner of the periodic table. Elements with high electronegativities tend to acquire electrons in chemical reactions and are found in the upper right corner of the periodic table. The most important method uses a measurement called electronegativity (represented by the Greek letter chi, χ, pronounced “ky” as in “sky”), defined as the relative ability of an atom to attract electrons to itself in a chemical compound. Conversely, the elements with the lowest ionization energies are generally those with the least negative electron affinities and are located in the lower left corner of the periodic table.īecause the tendency of an element to gain or lose electrons is so important in determining its chemistry, various methods have been developed to quantitatively describe this tendency. The elements with the highest ionization energies are generally those with the most negative electron affinities, which are located toward the upper right corner of the periodic table (compare Figure 2.9.2 and Figure 2.10.2). 
0 Comments
This Titanic movie is also having a unique tune that can touch your heart in various ways. Titanic is one of the biggest movies about a true incident, it is one of the best movies which is still watched around the globe. This is one of the most classic and melodious ringtones you can find for your mobile phones. Although you can get this original version of the Nokia ringtone in your current mobile phone by downloading it from the link provided below this article. As Nokia used to be the largest manufacturer of mobile devices in the world, it is most likely you have already used a Nokia device which is having an original Nokia ringtone. Nokia ringtone is one of the most trending ringtones to date, which is used in most of the mobile phones. Most of us think about the original Nokia ringtone if someone says ringtone. There are various versions of the Star Wars theme available for you to download, so we have provided the download link for you to download the original version of the Star Wars theme on your mobile phones. Get customers staying on your mobile phone and bring the essence of Star Wars each time someone calls on your mobile phone. In case you are a fan of Star Wars movies, then you will definitely be interested in the Star Wars theme ringtone. 
Luckily you're at home and your phone is ringing on an empty bus seat miles away.Star Wars is one of the biggest movie franchises available around the globe. Experimentation has taught you that other ringtones simply cause you to look around in bewilderment, thinking: "Where is that tinny version of the Mexican Hat Dance coming from?"Ħ5+: A terrible impression of Victor Meldrew saying: "I don't believe it!" You didn't want a phone, but one of your kids bought you one anyway, and they programmed this for you because you can be really grumpy sometimes, just like Victor Meldrew! You find it insufferable, but don't know how to change it. Using it ironically is problematic, not least because so many people still use it unironically.ĥ0-65: Old-fashioned telephone ring This is how a phone should sound. Match of the Day theme "I love football and haven't an original bone in my body."Ĥ0-50: Nokia tune The world's most recognisable ringtone, immortalised by Dom Joly in Trigger Happy TV. Answer by saying your own surname, or even just "yeah", followed by "affirmative, cat food is on my list".Ģ5-30: Nickelback's Rockstar You don't give a damn what anyone thinks about you, which is clearly a big part of what is wrong with you.ģ0-40: Mission: Impossible theme The CTU chirp of yesterday, though chances are you only remember the Tom Cruise movies, not the TV series. They're turning our weapons against us.Ģ0-25: CTU chirp By choosing the little sequence of beeps that Jack Bauer's phone produces whenever he receives a call on 24, you are telling the world, "Sorry, I have to take this, it could be of vital importance", even when you've spent the morning in Starbucks with a latte and a freesheet. This is the ultrasonic "Mosquito alarm" deployed to keep teens away from shops. 11-14: Catchphrases from Little Britain Only a schoolboy, or possibly my bank manager, could find a phone that says "Computer says no" indelibly comic.ġ4-20: Mosquito, AKA Teen Buzz Some ringtones are difficult to attribute to a specific age group, but not this one, because people over 25 can't hear it.  
|



 RSS Feed
RSS Feed
